|
People can be assured of the MHRA’s work to continuously monitor the tests in use as is our normal process.” This has allowed us to extend the EUA to allow ongoing supply of these LFDs over the coming months. “We have now concluded our review of the risk assessment and are satisfied that no further action is necessary or advisable at this time. A full risk assessment was undertaken by DHSC as legal manufacturer of the LFTs in the UK and the MHRA has undertaken a thorough review to ensure that we were satisfied with the assessment and any action proposed. Following our normal process to investigate any product concern, the MHRA immediately began reviewing all available information. MHRA director of devices Graeme Tunbridge told Medical Device Network: “Our priority is to ensure patients and the public have access to safe and effective medical devices and tests. This is because the MHRA has refused to disclose this information, despite freedom of information requests.” In their BMJ editorial, Pollock and Roderick said: “It is not known… what this review consisted of, what evidence the MHRA has as to whether the conditions of the original authorisation have been complied with and which justified the extension, why the extension is only for two months, and what the terms of the extended authorisation are. The MHRA conducted a review into the Innova tests following the US recall but reached a “satisfactory outcome” allowing the authorisation to be extended. The original six-month authorisation expired on 22 June but will now run until 28 August. Last month, the UK Medicines and Healthcare products Regulatory Agency (MHRA) announced that it had extended its exceptional use authorisation (EUA) for the Innova tests for just over two months. The tests it supplies are currently manufactured by Xiamen Biotime Biotechnology in Fujian, China. 
Innova is owned by Pasaca Capital, a California-based private equity firm, and is reportedly setting up a manufacturing plant in Wales. The DHSC has entered into eight different contracts with Innova Medical Group to acquire its LFTs, valued at £3.2bn as of May 2021. The performance characteristics of the test have not been adequately established, presenting a risk of false results.” Why Innova? The FDA said: “Labelling distributed with certain configurations of the test includes performance claims that did not accurately reflect the performance estimates observed during the clinical studies of the tests. The results of this study have been published just a month after the test was subject to a Class I recall in the US, with the Food and Drug Administration (FDA) advising citizens to “destroy the tests by placing them in the trash”. However, despite being cited by the government as evidence in favour of routine asymptomatic testing, the Liverpool study ultimately confirmed the low sensitivity of the tests in a mass asymptomatic screening setting.įinal data shows that while the test had an exceptionally high specificity of 99.94%, meaning false-positive rates were very low, the overall sensitivity was only 40%, meaning only four in ten of the 74 participants who tested positive by PCR would test positive using an LFT. The report also estimates that it led to a 21% reduction in cases until mid-December 2020, after which the Alpha variant surge made comparison difficult. The report estimates that the trial increased case detection by 18% compared to control areas. 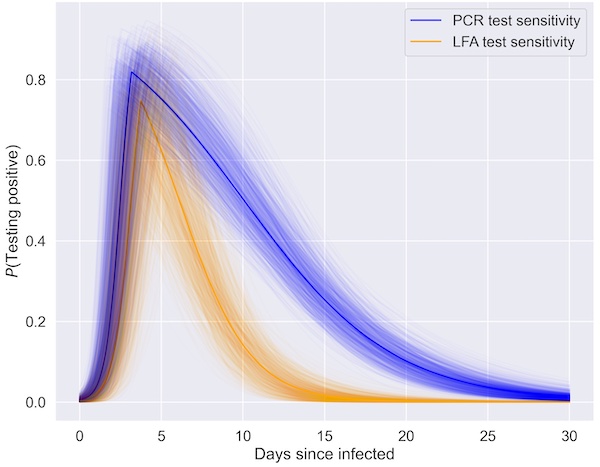
The pilot, which ran from 6-29 November 2020, saw the Innova SARS-CoV-2 antigen rapid lateral flow test offered to around 125,000 residents attending asymptomatic testing sites.Ī subset of 5,869 participants also provided samples for confirmatory polymerase chain reaction (PCR) tests. 
The third study the DHSC cited was the full evaluation of the Liverpool COVID-SMART community testing pilot for people without symptoms. However, for those with viral loads under 10,000 copies per mil, sensitivity dropped to only 43%. 
It found that for people with high sample viral loads above 1,000,000 copies per ml, LFTs were 96% effective, dropping slightly to 92% effective for individuals with a viral load between 10,000 – 1,000,000 copies per ml. The first study assessed the ability of the LFTs to detect the Delta and Alpha variants and found no change in the sensitivity of the tests when identifying either variant, results which scientists had largely been expecting already.Ī second study analysed the efficacy of LFTs under different testing conditions and varying patient viral loads by comparing a number of different LFT user evaluation studies, mostly of the Innova test. However, critics say that the devices’ apparent propensity for false-negative results will lead to infected people incorrectly assuming they are virus-free, potentially spreading the disease to others in the community.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
